Al atomic radius4/5/2023 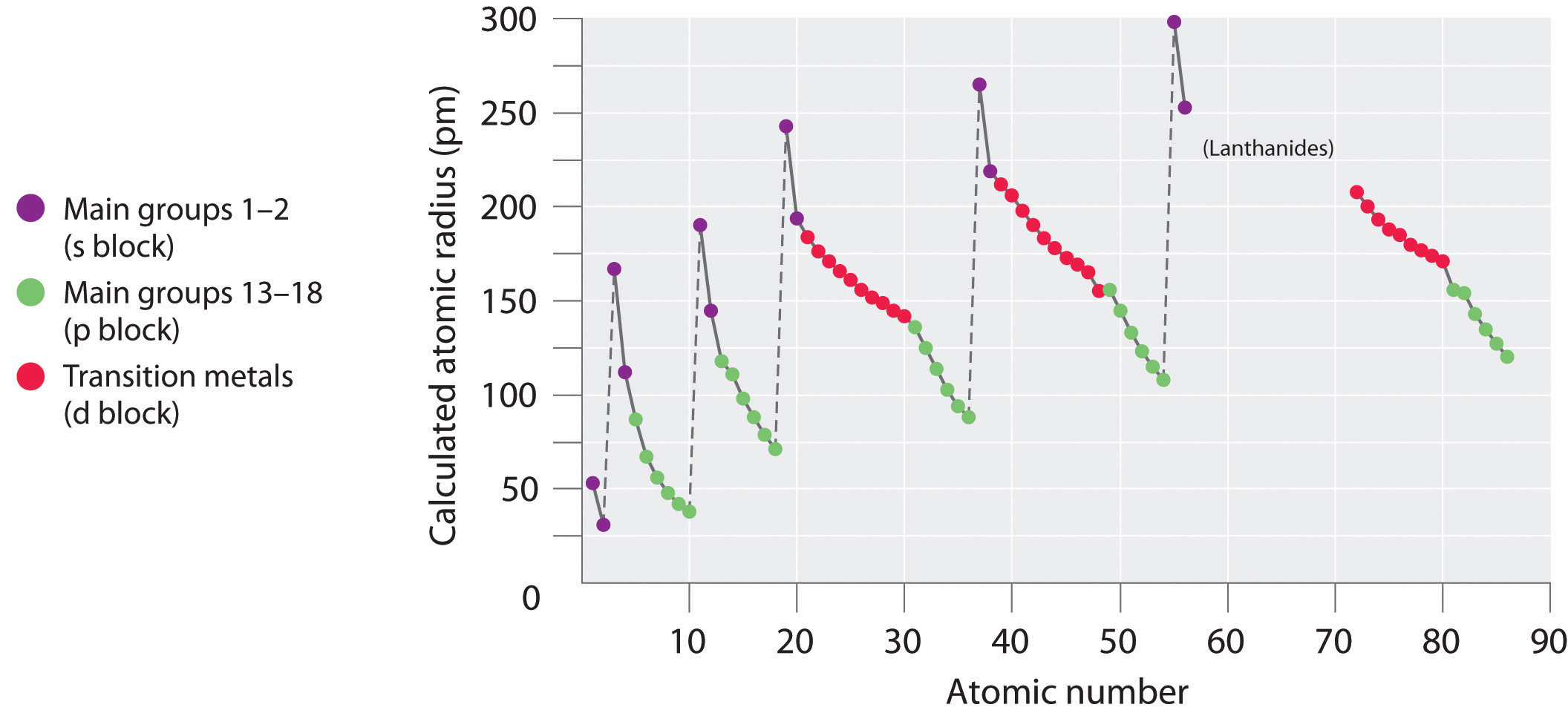
Moreover, an increasing Al content had a positive influence on the corrosion resistance of the alloy, increased corrosion potential of the alloy and reduced corrosion current. The atomic radius of rare earth element is 0.174-0.204 nm 12, which is larger than the atomic radius of aluminum (0.143 nm). According to the experimental results, the hardness increased by 30.4 %and the compressive strength increases by 22.9 %. Aluminium is a chemical element with atomic number 13 which means there are 13 protons in its nucleus. 
In addition, with the increase of Al content, the mechanical properties of the alloy are improved to some extent. Atomic Number Protons, Electrons and Neutrons in Aluminium. With the increase of Al content, the microstructure is gradually transformed into BCC structure, and the Cu segregation phenomenon is improved. The results show that when Al content is low, Cu segregation phenomenon is serious, and the microstructure of the alloy is FCC structure and a little BCC structure. The effects of the Al content on the microstructure, mechanical properties and corrosion behavior of CoCrCuFeNiMoAlx (x = 0, 0.32, 0.67, and 1.0) alloys were studied. For the phenomenon of Cu segregation, we report a strategy to tailor the existing form of Cu in HEA from undesired large-scale segregation to uniform distribution in the solid solution phase. Simply put, the atomic radius is half of the diameter of the atom, which is a result of the number of protons, neutrons, and electrons that compose the atom. Want to learn more details and data about Aluminum (Al)? Check my Elements Comprehensive List.A new multi-principal Co-Cr-Cu-Fe-Ni-Mo-Al high entropy alloy with good mechanical properties and corrosion resistance was prepared. Atomic radius of Oxygen (O) 152 pm: 9: Atomic radius of Fluorine (F) 135 pm: 10: Atomic radius of Neon (Ne) 154 pm: 11: Atomic radius of Sodium (Na) 227 pm: 12: Atomic radius of Magnesium (Mg) 173 pm: 13: Atomic radius of Aluminum (Al) 184 pm: 14: Atomic radius of Silicon (Si) 210 pm: 15: Atomic radius of Phosphorus (P) 180 pm: 16: Atomic. 
Third most abundant element in the earth's crust. Obtained by electrolysis from bauxite (Al2O3). Largest Smallest Answer Bank oxygen silicon calcium barium Arrange these elements according to atomic radius. The term 'atomic radius' itself is problematic: it may be. The value assigned to the radius of a particular atom will always depend on the definition chosen for 'atomic radius', and different definitions are more appropriate for different situations. This value may be the same as the atomic radius, or it may be larger for anions and the same. Largest radius Smallest radius Answer Bank Ar Si Mg Al Na Arrange the elements according to atomic radius, from largest to smallest. Atomic radius, and more generally the size of an atom, is not a precisely defined physical quantity, nor is it constant in all circumstances. The ionic radius is half the distance between two gas atoms that are just touching each other. In other words, it is half the diameter of an atom, measuring across the outer stable electrons. Too soft in its pure form so less than 1% of silicon or iron is added, which hardens and strengthens it. The atomic radius is half the diameter of a neutral atom. This site offers comprehensive information for each element including: who, when & where up to 40 properties (chemical & physical) over 3,600 nuclides (isotopes) over 4,400 nuclide decay modes the element names in 10 different languages and more. Used for many purposes from airplanes to beverage cans. In the case of Aluminum the atomic volume is 10.0 cm³/mol. There are interesting things about Aluminum that people don't know about. The atomic radius of an element tends to increase the further down you go in an element group. Note: Learn more about the molar (or atomic) volume here. No matter what criteria you use to describe the atomic radius, the size of an atom is dependent on how far out its electrons extend. Ok, so what is the atomic radius of Aluminum? The atomic radius of Ga is less than that of Al (Atomic radius of Al is 143 pm while that of Ga is 135 pm.). Assuming the end displacement of an end-loaded cantilevered beam is given. Relative changes in the mean squared nuclear charge distribution can be precisely measured with atomic spectroscopy. Wong et al 48 measured the bending force of MWCNTs using atomic force microscopy. It can be measured by the scattering of electrons by the nucleus. Atoms have a theoretical atomic volume, even Aluminum. On moving down the group, for each successive element, one extra shell of electrons is added and, hence, atomic radius is expected to increase. The rms charge radius is a measure of the size of an atomic nucleus, particularly the proton distribution.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
